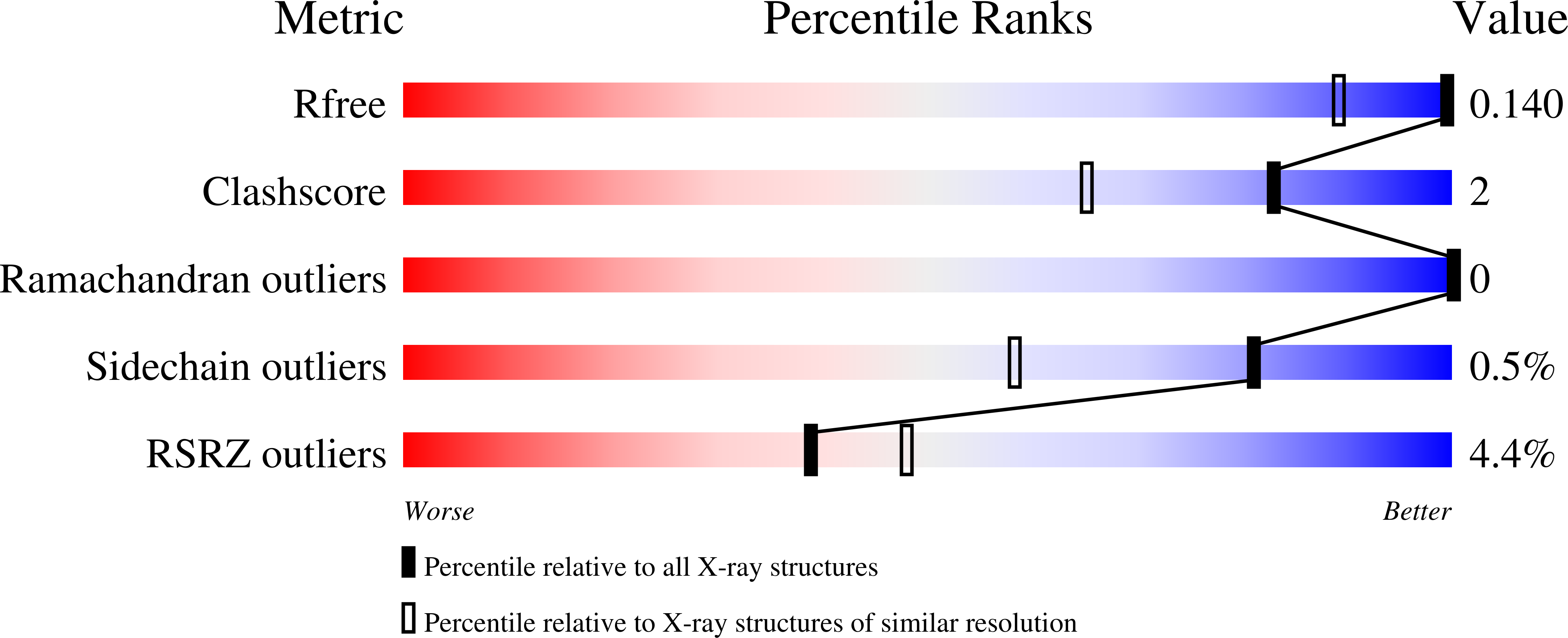
Structural Basis for Substrate Recognition and Hydrolysis by Mouse Carnosinase CN2* - Journal of Biological Chemistry

A QM/MM simulation study of transamination reaction at the active site of aspartate aminotransferase: Free energy landscape and proton transfer pathways - Dutta Banik - 2020 - Journal of Computational Chemistry - Wiley Online Library
Structures of the apo- and the metal ion-activated forms of the diphtheria tox repressor from Corynebacterium diphtheriae. - Abstract - Europe PMC
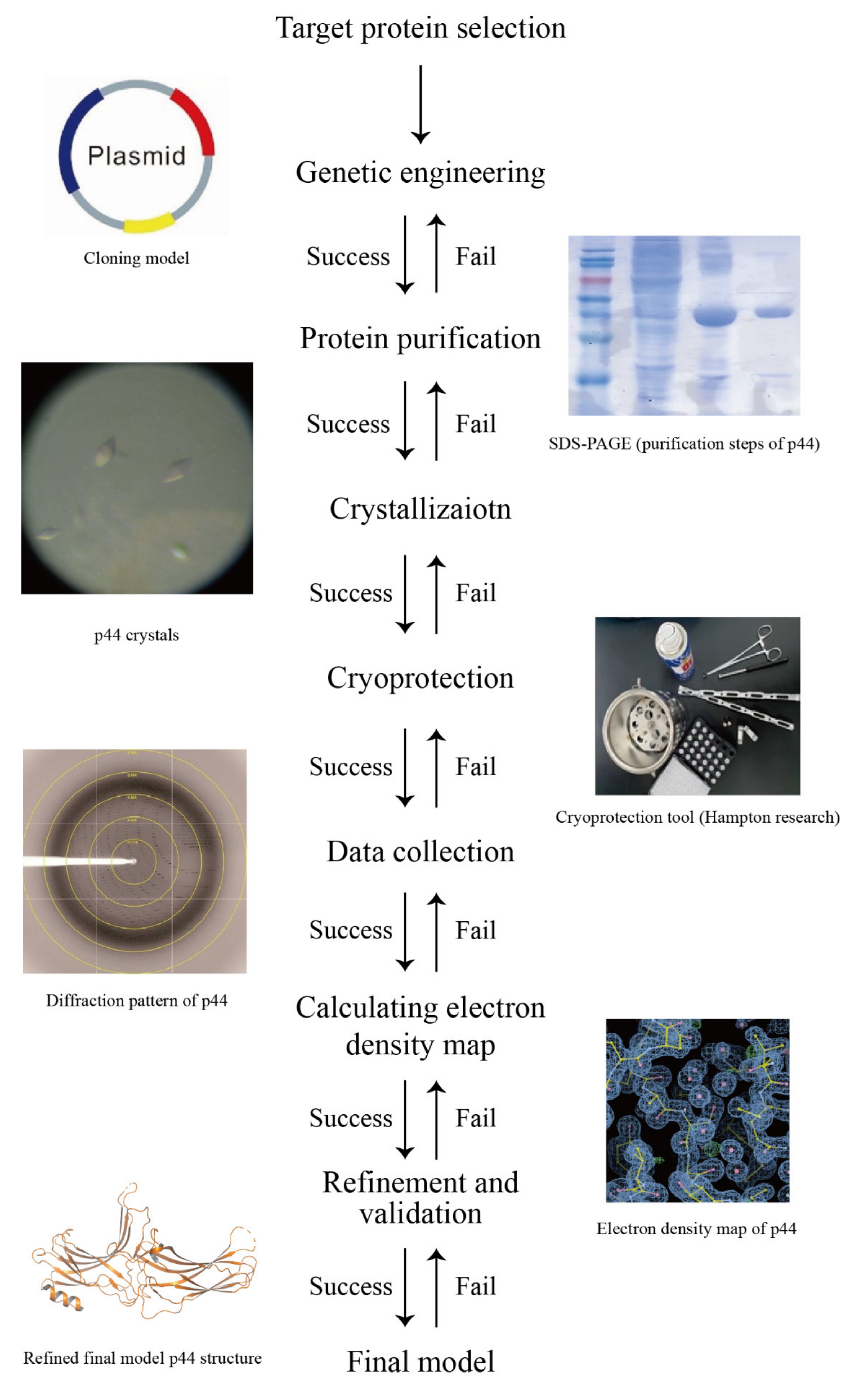
Crystals | Free Full-Text | A Short Review on Cryoprotectants for 3D Protein Structure Analysis | HTML
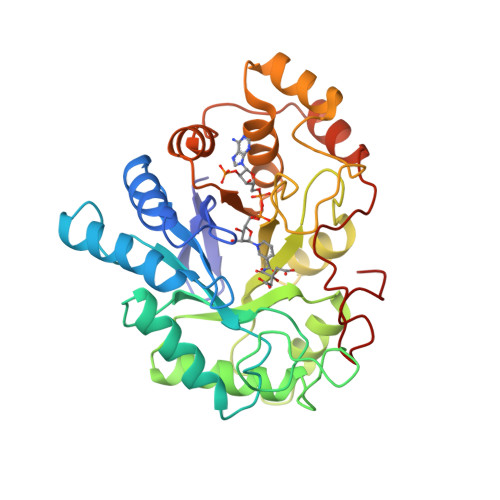
2ACU: TYROSINE-48 IS THE PROTON DONOR AND HISTIDINE-110 DIRECTS SUBSTRATE STEREOCHEMICAL SELECTIVITY IN THE REDUCTION REACTION OF HUMAN ALDOSE REDUCTASE: ENZYME KINETICS AND THE CRYSTAL ... - RCSB PDB